Who We Are
Medical Equipment Manufacturing with Practical Export Support
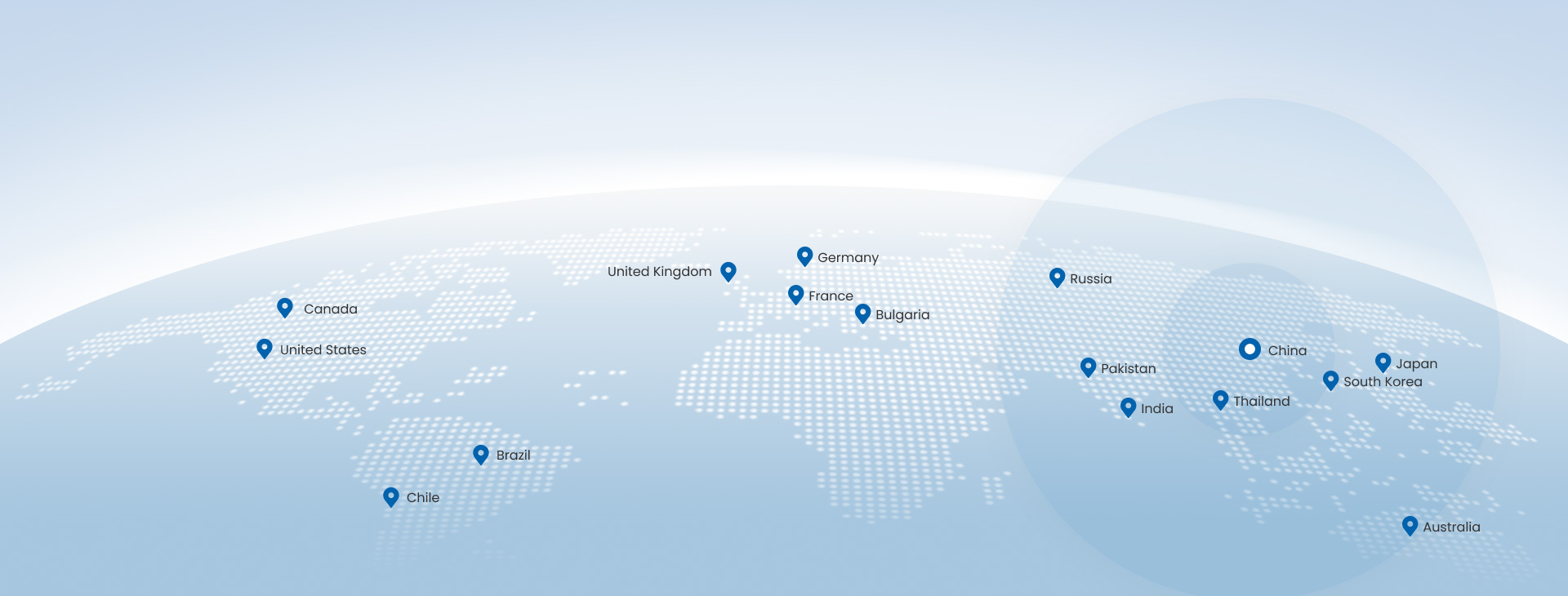
Shandong Yuda Medical Device Technology Group Co., Ltd. provides operating room equipment and related hospital solutions, including operating tables, obstetric tables, examination chairs, medical pendant systems, surgical lights, traction frames, positioning gel pads, operating table accessories, medical carts, and hospital beds. We support OEM/ODM projects and distributor partnerships with documented QC, compliance-focused manufacturing, and export-ready packaging.
Our core team brings 20+ years of industry experience in medical equipment manufacturing, quality control, and export support. This experience is reflected in product selection guidance, configuration advice, compliance communication, and after-sales coordination for international buyers.
For verification, please review our certifications, explore our factory tour, or contact our team for a product dossier and compliance documents.